
10 October, 2024

Author - Barbara De Kegel (left). Dr Colm Ryan (right).
Posted: 29 September, 2021
Understanding how genetic mutations alter tumour cells is vital to cancer research and future drug development. Barbara De Kegel, a PhD student funded by the Irish Research Council under a Laureate Award awarded to Dr Colm Ryan at Systems Biology Ireland and the School of Computer Science at UCD, is the lead author on a paper that presents a new machine learning model for complex genetic analysis. Published in the journal Cell Systems, the paper demonstrates the research team’s development of a computational tool that identifies vulnerabilities in tumour cells that could be therapeutically exploited.
Targeted therapies, which aim to selectively kill cancer cells while limiting toxicity to healthy cells, are based on exploiting specific vulnerabilities that are present in a patient’s tumour. One means to identify such vulnerabilities is to pinpoint genetic mutations that occur in tumour cells and distinguish them from healthy cells. In some cases loss of one gene, through a mutation or deletion, can make tumours particularly vulnerable to the loss of a second gene – this concept is known as synthetic lethality.
Synthetic lethality can occur when genes have (partially) redundant functionality and are thus individually dispensable but jointly required for the tumour cell’s survival. One major source of redundancy in our genome is gene duplication – over half of our genes were duplicated at some point in evolutionary history, resulting in duplicate genes called paralogs. Although the genes making up a paralog pair are rarely perfect copies of each other, many are able to compensate for each other’s loss. In this way paralogs can be viewed as “spare tires” for the cell. If we know a tumour cell has lost one member of a paralog pair (flat tire), we might anticipate that it will be especially vulnerable to the loss of the other member (the spare tire).
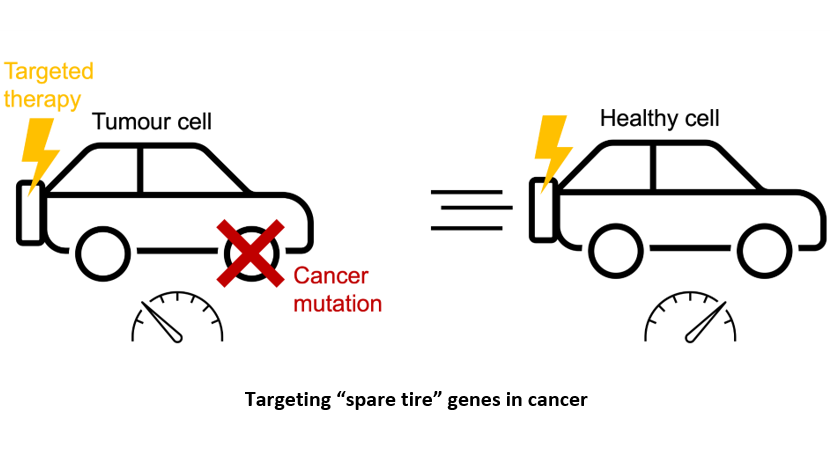
Given that over 60% of our genes have at least one paralog, exploiting synthetic lethality between paralog pairs could be a widely applicable therapeutic approach. This is especially the case for synthetic lethalities that involve a paralog that is frequently lost in cancer, as many patients’ tumours would then be reliant on the same “spare tire”, making it an attractive drug target. However, not all paralog pairs are synthetic lethal and testing them is slow and expensive – to date only about 5% of paralog pairs have been tested. In addition, it is not clear which factors make specific paralog pairs likely to be synthetic lethal.
In a new study published in Cell Systems, we developed a computational tool to facilitate the discovery and understanding of synthetic lethal paralog pairs. Our tool uses machine learning to make predictions for over 36,000 paralog pairs, resulting in a ranking of which pairs are most likely to be synthetic lethal. We also used an approach from the field of explainable artificial intelligence to provide explanations for each individual prediction. This showed us that paralog pairs can be synthetic lethal for different reasons – in some cases this was due to the second gene being very functionally similar to the first, while in other cases this was more due to the first gene being highly essential for the functioning of the cell.
We used our predictions to identify new synthetic lethal relationships involving paralogs that are frequently lost in cancer. We validated two of these synthetic lethal relationships experimentally in the lab, including one that involves a gene that is recurrently lost in prostate cancer. These relationships can serve as potential starting points for new drug discovery efforts. In addition all our predictions along with their explanations have been made publicly available at cancergd.org/static/paralogs, so that they can be used by other research groups or pharmaceutical companies as a resource for prioritizing which paralog pairs to investigate further in the search for novel therapeutic targets.
Barbara De Kegel is funded by the Irish Research Council under a Laureate Award awarded to Dr Colm Ryan and is based in Systems Biology Ireland and the School of Computer Science at UCD.
The opinions expressed here are those of the author and do not represent or reflect the views of the Irish Research Council.